|
The reactivity series tells whether a metal can displace another metal in a displacement reaction or not. It is generally a vertically presented series that contains the most-reactive elements at the top of the series and the least-reactive elements at the bottom. 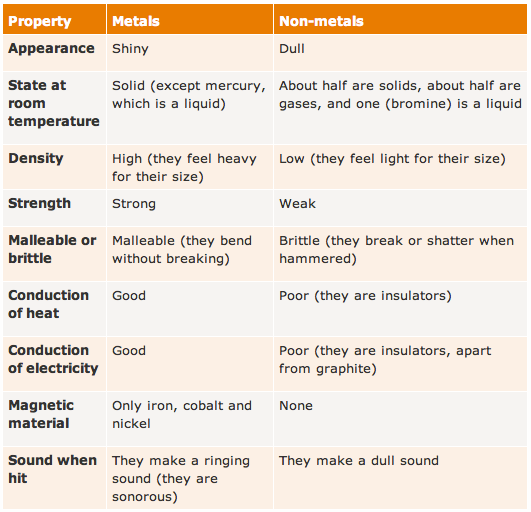
So, it is a list of metals arranged in order of decreasing reactivity. It means the more reactive metals come first in this series followed by less reactive metals. In this series, the metals are arranged in descending order of their reactivity. Knowledge of chemistry is endless, so to know more about various types of chemical reactions.It refers to the reactivity series of metals or elements which are metals. Therefore, nonmetals that gain electrons rapidly are more active than others. We determine the reactivity of nonmetals, upon their capability to gain electrons in the solution state to form negatively charged ions or anions. What determines the reactivity of nonmetals?Īns. Therefore, the more readily metal loses its electrons, the more active it is, and higher up it is in the reactivity series. We determine the reactivity of metals, upon their capability to lose electrons in the solution state to form positive ions or cations. What determines the reactivity of metals?Īns. Fluorine is the most active nonmetals and it displaces all other nonmetals in salt solutions. Which is the most active or reactive nonmetal?Īns. Due to its low reactivity, it found uses in the manufacture of laboratory equipment, electrodes, platinum resistance thermometers, dentistry equipment, and jewelry. It has remarkable resistance to corrosion, even at high temperatures, and is therefore considered a noble metal. Platinum is one of the least reactive metals and therefore lies at the bottom of the reactivity series. Which is the least active or reactive metal?Īns. The table below, explain the reactions of metals with oxygen (air), water, dilute acids, and other salt solutions, at various level of the reactivity series.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
